Why Is Pancreatic Cancer So Deadly? The Science Is Finally Changing
By: Joy Stephenson-Laws, JD, Founder
When Michael Landon was diagnosed with pancreatic cancer in April 1991, he was dead by July. Three months. Patrick Swayze was diagnosed at stage IV in 2008 and lived twenty months — long enough to finish a television series, but not long enough to see his sixtieth birthday. Sally Ride, the first American woman in space, survived seventeen months. Alan Rickman kept his diagnosis private and was gone within a year. Alex Trebek made his fight public and bought himself twenty months of clarity, work, and goodbyes.
The list is long, and the pattern is unmistakable: pancreatic cancer kills quickly, and it kills people who, by every other measure, should have had decades left. To understand why, you have to understand the pancreas itself — what it does, how cancer hijacks it, and why this disease has resisted treatment in ways that breast, colon, and even lung cancer have not.
That last part is starting to change. But the change only makes sense once you understand what the pancreas is up against.
What does the pancreas do? Two organs in one
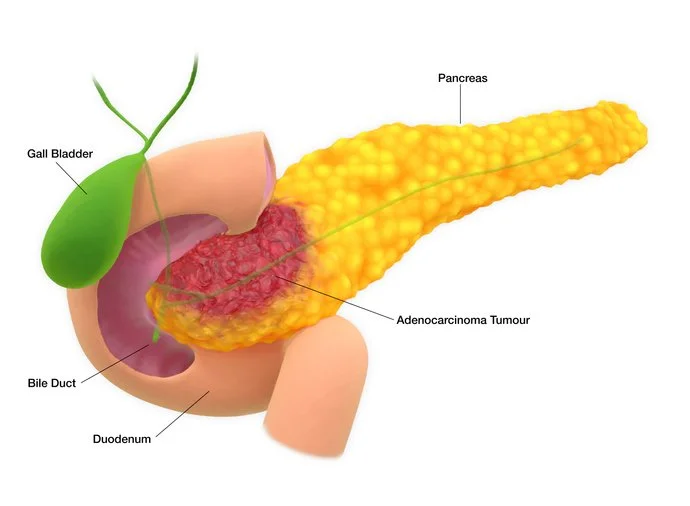
The pancreas is a six-inch organ tucked behind the stomach, deep in the abdomen, with two completely separate jobs.
The exocrine pancreas — about 95% of the organ's tissue — produces digestive enzymes. Every time you eat, the pancreas secretes lipase, amylase, and proteases through a duct that empties into the small intestine. Without these enzymes, you cannot break down fat, starch, or protein. People who lose pancreatic function develop greasy, foul-smelling stools (steatorrhea) and lose weight no matter how much they eat.
The endocrine pancreas — the remaining 5% — is a scattered cluster of hormone-producing cells called islets of Langerhans. These cells make insulin, glucagon, and several other hormones that regulate blood sugar. When islet cells fail, you get diabetes.
This dual-organ structure matters enormously, because pancreatic cancer is not one disease. It's two.
Types of pancreatic cancer: PDAC vs. neuroendocrine tumors
About 95% of pancreatic cancers arise from the exocrine cells, almost always from the lining of the ducts. This is pancreatic ductal adenocarcinoma, or PDAC. It is what people mean when they say "pancreatic cancer," and it is one of the most lethal malignancies in medicine. The 5-year relative survival rate for all stages combined is about 13%.
The other 5% are pancreatic neuroendocrine tumors (PNETs), which arise from the hormone-producing islet cells. These behave very differently. They tend to grow more slowly, are often caught earlier, and respond to different treatments. Steve Jobs had a PNET. So did Aretha Franklin. When you see headlines that say a celebrity "battled pancreatic cancer for years," they almost always had a PNET — because surviving PDAC for years is rare.
This is not a pedantic distinction. Conflating the two creates false hope on one end and unfair fear on the other. The rest of this article is about PDAC, because that is the disease driving the survival statistics and the death toll.
Why is pancreatic cancer so deadly? 4 biological reasons
PDAC is not deadly because doctors aren't trying. It's deadly because the tumor's biology is uniquely hostile.
It hides. The pancreas sits retroperitoneally — behind the stomach, alongside the spine, surrounded by other organs. A tumor can grow for months or years without causing symptoms severe enough to investigate. When symptoms do appear, they are often vague: dull abdominal pain, mid-back pain, fatigue, mild weight loss. By the time something specific shows up — jaundice, sudden-onset diabetes, dramatic weight loss — the tumor has often spread. Only about 15% of PDAC cases are caught while still localized to the pancreas. Most are diagnosed when the cancer has already reached lymph nodes (regional) or distant organs like the liver and lungs (distant).
Stage at diagnosis dictates survival, ruthlessly. According to SEER data, the 5-year relative survival rate is around 44% for localized disease, 16% for regional disease, and 3% for distant disease. The single most important variable in pancreatic cancer outcomes is whether the cancer is found before it leaves the pancreas — and most of the time, it isn't.
It is encased in armor. PDAC tumors surround themselves with an unusually dense layer of scar-like tissue called desmoplastic stroma. This fibrous shell physically blocks chemotherapy drugs from reaching the cancer cells, suppresses the local immune response, and creates a low-oxygen, low-nutrient microenvironment that selects for the most aggressive tumor cells. Drugs that work in other cancers often can't even get to the target.
It is driven by a mutation no one could drug. More than 90% of PDAC tumors carry mutations in a gene called KRAS. KRAS makes a protein that sits on the inside of the cell membrane and tells the cell when to grow. Mutated KRAS gets stuck in the "on" position, driving uncontrolled growth. For four decades, drug developers tried to block KRAS and failed — the protein's surface is smooth, with no obvious pocket for a drug to grab onto. KRAS earned a reputation as the "undruggable" target.
That reputation is now breaking.
Pancreatic cancer treatment breakthroughs in 2026
In April 2026, Revolution Medicines reported Phase 3 trial results for a once-daily pill called daraxonrasib that targets a broad range of mutated RAS proteins. In patients with previously treated metastatic PDAC — a narrow but historically untreatable population — those who took the drug lived a median of 13.2 months versus 6.7 months on chemotherapy alone, with a 60% reduction in the risk of death. The principal investigator called the results practice-changing. (These are topline results from a company press release; full data is expected at the 2026 ASCO meeting, and the relevant comparison is to chemotherapy in this specific second-line population.)
A day earlier, Northwestern researchers published Phase 2 results in Nature Medicine for a different drug, elraglusib, which targets the tumor microenvironment rather than the cancer cells themselves. Patients on elraglusib plus standard chemotherapy were twice as likely to be alive at one year, with a 38% reduction in the risk of death.
Memorial Sloan Kettering reported additional KRAS-targeted results at AACR in April, including early data on a first-in-class KRAS degrader — a drug that doesn't just block the mutant protein but tags it for destruction.
None of this cures pancreatic cancer. Phase 2 results need Phase 3 confirmation. Most patients still don't qualify for these therapies. But after decades of failed trials, the convergence is real: the biology that made PDAC untreatable is, finally, yielding.
Pancreatic cancer risk factors, warning signs, and what you can do
Awareness without action is just anxiety. Here is what is supported by evidence.
Reduce modifiable risk. Smoking roughly doubles pancreatic cancer risk and accounts for about 25% of cases. Stopping smoking is the single highest-impact lifestyle change. Obesity, heavy alcohol use, and poorly controlled type 2 diabetes are all independent risk factors. Chronic pancreatitis — repeated inflammation of the pancreas, often from heavy alcohol use — substantially raises risk over time.
Know the disparities, and your own family history. Black Americans are diagnosed with pancreatic cancer at higher rates than white Americans — roughly 20% higher in adjusted analyses, with some datasets showing larger gaps — and have worse outcomes, driven by a combination of access, comorbidities, and biological factors that are still being studied. Family history matters, too: about 10% of PDAC cases are linked to inherited mutations. BRCA2 mutations (best known for breast and ovarian cancer risk) raise pancreatic cancer lifetime risk roughly two- to threefold above the general population baseline; BRCA1 mutations carry a smaller but still elevated risk. Other hereditary syndromes — Lynch, Peutz-Jeghers, FAMMM, hereditary pancreatitis, ATM, PALB2 — also confer increased risk, with hereditary pancreatitis carrying the highest lifetime risk (25–40%). People of Ashkenazi Jewish ancestry have a higher prevalence of BRCA2 mutations. If you have two or more first-degree relatives with pancreatic cancer, or a known inherited mutation, talk to a genetic counselor.
Take new-onset diabetes after age 50 seriously. Diabetes that appears suddenly in someone over 50 with no obvious risk factors — especially when paired with weight loss — can be an early sign of pancreatic cancer. It is not a reason to panic, but it is a reason to ask your doctor whether further evaluation is warranted.
Pancreatic cancer symptoms you should not ignore
Painless jaundice (yellowing of the skin or eyes), pale stools, dark urine, mid-back pain that wraps around to the abdomen, sudden unexplained weight loss, and new digestive changes that don't resolve — these warrant evaluation, not reassurance. Most of the time the cause is benign. But "most of the time" is not all of the time.
Pancreatic cancer screening for high-risk individuals: what surveillance looks like
Surveillance is not abstract for me. I have a genetic predisposition to pancreatic cancer and a pancreatic cyst under watch, which is why I pay particular attention to how the evidence on this is evolving — both for my own care and for other high-risk patients who are proactive about their health and follow us here at Proactive Health Labs. So this section is the one I most want to get right.
There is no validated screening test for pancreatic cancer in the general population. CA 19-9 is a tumor marker, not a screening test, and using it that way produces too many false positives and false negatives to be useful.
For high-risk individuals, however, surveillance is real, recommended, and increasingly evidence-based. The International Cancer of the Pancreas Screening (CAPS) Consortium and the National Comprehensive Cancer Network (NCCN) recommend annual surveillance with MRI/MRCP and/or endoscopic ultrasound (EUS) — alternating between the two, with neither using ionizing radiation — for people who meet specific criteria. Those criteria generally include carriers of BRCA1, BRCA2, ATM, PALB2, CDKN2A, STK11 (Peutz-Jeghers), and Lynch syndrome mutations, particularly those with a family history of pancreatic cancer. Recommended starting ages vary by mutation: age 50 (or ten years before the earliest family case) for most, age 40 for CDKN2A and hereditary pancreatitis, age 30–35 for Peutz-Jeghers.
The evidence for this approach is now strong enough to matter. The combined CAPS1–5 cohort (Dbouk, Canto et al., Journal of Clinical Oncology 2022; updated analysis Blackford, Canto et al., JAMA Oncology 2024) followed 1,731 high-risk individuals across more than two decades of surveillance. Of 26 PDAC cases identified across the program, 19 were detected during active surveillance — and of those screen-detected cancers, 57.9% were caught at stage I, with another 15.8% at stage II. The 5-year survival rate among screen-detected cases was 73.3%, with a median overall survival of 9.8 years. The matched-cohort analysis published in JAMA Oncology compared all 26 high-risk individuals with PDAC against 1,504 SEER controls and found 5-year overall survival of 50% versus 9% — a fourfold improvement. By contrast, six of seven cases found outside surveillance (in patients who had stopped their annual screenings) were stage IV, with a median survival of 1.5 years.
That is the difference surveillance can make in the right population.
If you have a family history that concerns you, or a known mutation, the right move is a referral to a pancreatic surveillance program at a high-volume center. Don't try to assemble this on your own.
The bottom line on pancreatic cancer survival
Pancreatic cancer remains one of the deadliest cancers in medicine, and pretending otherwise does no one any favors. The reasons are biological, not bureaucratic — late detection, dense stroma, and a driver mutation that resisted forty years of drug development. But the same science that mapped those obstacles has, in the past two years, produced the first treatments to meaningfully extend survival in advanced disease. And in parallel, surveillance is finally letting some high-risk individuals catch the disease early enough to do something about it.
For everyone else, the most powerful tool is the same one it has always been: pay attention to your body, know your family history, and don't let "vague" become an excuse for "ignored." The patients who do best are still, overwhelmingly, the ones whose disease is found early. Everything else — the new drugs, the new science, the new optimism — is built on that foundation.
Why Is Pancreatic Cancer So Deadly? The Science Is Finally Changing
Meta description: Pancreatic cancer remains one of medicine's deadliest diseases — but new RAS-targeted treatments and surveillance programs are finally changing outcomes in 2026. Here's why the disease is so hard to treat, and what's working now.
When Michael Landon was diagnosed with pancreatic cancer in April 1991, he was dead by July. Three months. Patrick Swayze was diagnosed at stage IV in 2008 and lived twenty months — long enough to finish a television series, but not long enough to see his sixtieth birthday. Sally Ride, the first American woman in space, survived seventeen months. Alan Rickman kept his diagnosis private and was gone within a year. Alex Trebek made his fight public and bought himself twenty months of clarity, work, and goodbyes.
The list is long, and the pattern is unmistakable: pancreatic cancer kills quickly, and it kills people who, by every other measure, should have had decades left. To understand why, you have to understand the pancreas itself — what it does, how cancer hijacks it, and why this disease has resisted treatment in ways that breast, colon, and even lung cancer have not.
That last part is starting to change. But the change only makes sense once you understand what the pancreas is up against.
What does the pancreas do? Two organs in one
The pancreas is a six-inch organ tucked behind the stomach, deep in the abdomen, with two completely separate jobs.
The exocrine pancreas — about 95% of the organ's tissue — produces digestive enzymes. Every time you eat, the pancreas secretes lipase, amylase, and proteases through a duct that empties into the small intestine. Without these enzymes, you cannot break down fat, starch, or protein. People who lose pancreatic function develop greasy, foul-smelling stools (steatorrhea) and lose weight no matter how much they eat.
The endocrine pancreas — the remaining 5% — is a scattered cluster of hormone-producing cells called islets of Langerhans. These cells make insulin, glucagon, and several other hormones that regulate blood sugar. When islet cells fail, you get diabetes.
This dual-organ structure matters enormously, because pancreatic cancer is not one disease. It's two.
Types of pancreatic cancer: PDAC vs. neuroendocrine tumors
About 95% of pancreatic cancers arise from the exocrine cells, almost always from the lining of the ducts. This is pancreatic ductal adenocarcinoma, or PDAC. It is what people mean when they say "pancreatic cancer," and it is one of the most lethal malignancies in medicine. The 5-year relative survival rate for all stages combined is about 13%.
The other 5% are pancreatic neuroendocrine tumors (PNETs), which arise from the hormone-producing islet cells. These behave very differently. They tend to grow more slowly, are often caught earlier, and respond to different treatments. Steve Jobs had a PNET. So did Aretha Franklin. When you see headlines that say a celebrity "battled pancreatic cancer for years," they almost always had a PNET — because surviving PDAC for years is rare.
This is not a pedantic distinction. Conflating the two creates false hope on one end and unfair fear on the other. The rest of this article is about PDAC, because that is the disease driving the survival statistics and the death toll.
Why is pancreatic cancer so deadly? 4 biological reasons
PDAC is not deadly because doctors aren't trying. It's deadly because the tumor's biology is uniquely hostile.
It hides. The pancreas sits retroperitoneally — behind the stomach, alongside the spine, surrounded by other organs. A tumor can grow for months or years without causing symptoms severe enough to investigate. When symptoms do appear, they are often vague: dull abdominal pain, mid-back pain, fatigue, mild weight loss. By the time something specific shows up — jaundice, sudden-onset diabetes, dramatic weight loss — the tumor has often spread. Only about 15% of PDAC cases are caught while still localized to the pancreas. Most are diagnosed when the cancer has already reached lymph nodes (regional) or distant organs like the liver and lungs (distant).
Stage at diagnosis dictates survival, ruthlessly. According to SEER data, the 5-year relative survival rate is around 44% for localized disease, 16% for regional disease, and 3% for distant disease. The single most important variable in pancreatic cancer outcomes is whether the cancer is found before it leaves the pancreas — and most of the time, it isn't.
It is encased in armor. PDAC tumors surround themselves with an unusually dense layer of scar-like tissue called desmoplastic stroma. This fibrous shell physically blocks chemotherapy drugs from reaching the cancer cells, suppresses the local immune response, and creates a low-oxygen, low-nutrient microenvironment that selects for the most aggressive tumor cells. Drugs that work in other cancers often can't even get to the target.
It is driven by a mutation no one could drug. More than 90% of PDAC tumors carry mutations in a gene called KRAS. KRAS makes a protein that sits on the inside of the cell membrane and tells the cell when to grow. Mutated KRAS gets stuck in the "on" position, driving uncontrolled growth. For four decades, drug developers tried to block KRAS and failed — the protein's surface is smooth, with no obvious pocket for a drug to grab onto. KRAS earned a reputation as the "undruggable" target.
That reputation is now breaking.
Pancreatic cancer treatment breakthroughs in 2026
In April 2026, Revolution Medicines reported Phase 3 trial results for a once-daily pill called daraxonrasib that targets a broad range of mutated RAS proteins. In patients with previously treated metastatic PDAC — a narrow but historically untreatable population — those who took the drug lived a median of 13.2 months versus 6.7 months on chemotherapy alone, with a 60% reduction in the risk of death. The principal investigator called the results practice-changing. (These are topline results from a company press release; full data is expected at the 2026 ASCO meeting, and the relevant comparison is to chemotherapy in this specific second-line population.)
A day earlier, Northwestern researchers published Phase 2 results in Nature Medicine for a different drug, elraglusib, which targets the tumor microenvironment rather than the cancer cells themselves. Patients on elraglusib plus standard chemotherapy were twice as likely to be alive at one year, with a 38% reduction in the risk of death.
Memorial Sloan Kettering reported additional KRAS-targeted results at AACR in April, including early data on a first-in-class KRAS degrader — a drug that doesn't just block the mutant protein but tags it for destruction.
None of this cures pancreatic cancer. Phase 2 results need Phase 3 confirmation. Most patients still don't qualify for these therapies. But after decades of failed trials, the convergence is real: the biology that made PDAC untreatable is, finally, yielding.
Pancreatic cancer risk factors, warning signs, and what you can do
Awareness without action is just anxiety. Here is what is supported by evidence.
Reduce modifiable risk. Smoking roughly doubles pancreatic cancer risk and accounts for about 25% of cases. Stopping smoking is the single highest-impact lifestyle change. Obesity, heavy alcohol use, and poorly controlled type 2 diabetes are all independent risk factors. Chronic pancreatitis — repeated inflammation of the pancreas, often from heavy alcohol use — substantially raises risk over time.
Know the disparities, and your own family history. Black Americans are diagnosed with pancreatic cancer at higher rates than white Americans — roughly 20% higher in adjusted analyses, with some datasets showing larger gaps — and have worse outcomes, driven by a combination of access, comorbidities, and biological factors that are still being studied. Family history matters, too: about 10% of PDAC cases are linked to inherited mutations. BRCA2 mutations (best known for breast and ovarian cancer risk) raise pancreatic cancer lifetime risk roughly two- to threefold above the general population baseline; BRCA1 mutations carry a smaller but still elevated risk. Other hereditary syndromes — Lynch, Peutz-Jeghers, FAMMM, hereditary pancreatitis, ATM, PALB2 — also confer increased risk, with hereditary pancreatitis carrying the highest lifetime risk (25–40%). People of Ashkenazi Jewish ancestry have a higher prevalence of BRCA2 mutations. If you have two or more first-degree relatives with pancreatic cancer, or a known inherited mutation, talk to a genetic counselor.
Take new-onset diabetes after age 50 seriously. Diabetes that appears suddenly in someone over 50 with no obvious risk factors — especially when paired with weight loss — can be an early sign of pancreatic cancer. It is not a reason to panic, but it is a reason to ask your doctor whether further evaluation is warranted.
Pancreatic cancer symptoms you should not ignore
Painless jaundice (yellowing of the skin or eyes), pale stools, dark urine, mid-back pain that wraps around to the abdomen, sudden unexplained weight loss, and new digestive changes that don't resolve — these warrant evaluation, not reassurance. Most of the time the cause is benign. But "most of the time" is not all of the time.
Pancreatic cancer screening for high-risk individuals: what surveillance looks like
Surveillance is not abstract for me. I have a genetic predisposition to pancreatic cancer and a pancreatic cyst under watch, which is why I pay particular attention to how the evidence on this is evolving — both for my own care and for other high-risk patients who are proactive about their health and follow us here at Proactive Health Labs. So this section is the one I most want to get right.
There is no validated screening test for pancreatic cancer in the general population. CA 19-9 is a tumor marker, not a screening test, and using it that way produces too many false positives and false negatives to be useful.
For high-risk individuals, however, surveillance is real, recommended, and increasingly evidence-based. The International Cancer of the Pancreas Screening (CAPS) Consortium and the National Comprehensive Cancer Network (NCCN) recommend annual surveillance with MRI/MRCP and/or endoscopic ultrasound (EUS) — alternating between the two, with neither using ionizing radiation — for people who meet specific criteria. Those criteria generally include carriers of BRCA1, BRCA2, ATM, PALB2, CDKN2A, STK11 (Peutz-Jeghers), and Lynch syndrome mutations, particularly those with a family history of pancreatic cancer. Recommended starting ages vary by mutation: age 50 (or ten years before the earliest family case) for most, age 40 for CDKN2A and hereditary pancreatitis, age 30–35 for Peutz-Jeghers.
The evidence for this approach is now strong enough to matter. The combined CAPS1–5 cohort (Dbouk, Canto et al., Journal of Clinical Oncology 2022; updated analysis Blackford, Canto et al., JAMA Oncology 2024) followed 1,731 high-risk individuals across more than two decades of surveillance. Of 26 PDAC cases identified across the program, 19 were detected during active surveillance — and of those screen-detected cancers, 57.9% were caught at stage I, with another 15.8% at stage II. The 5-year survival rate among screen-detected cases was 73.3%, with a median overall survival of 9.8 years. The matched-cohort analysis published in JAMA Oncology compared all 26 high-risk individuals with PDAC against 1,504 SEER controls and found 5-year overall survival of 50% versus 9% — a fourfold improvement. By contrast, six of seven cases found outside surveillance (in patients who had stopped their annual screenings) were stage IV, with a median survival of 1.5 years. That is the difference surveillance can make in the right population.
If you have a family history that concerns you, or a known mutation, the right move is a referral to a pancreatic surveillance program at a high-volume center. Don't try to assemble this on your own.
The bottom line on pancreatic cancer survival
Pancreatic cancer remains one of the deadliest cancers in medicine, and pretending otherwise does no one any favors. The reasons are biological, not bureaucratic — late detection, dense stroma, and a driver mutation that resisted forty years of drug development. But the same science that mapped those obstacles has, in the past two years, produced the first treatments to meaningfully extend survival in advanced disease. And in parallel, surveillance is finally letting some high-risk individuals catch the disease early enough to do something about it.
For everyone else, the most powerful tool is the same one it has always been: pay attention to your body, know your family history, and don't let "vague" become an excuse for "ignored." The patients who do best are still, overwhelmingly, the ones whose disease is found early. Everything else — the new drugs, the new science, the new optimism — is built on that foundation.
References
Survival, incidence, and stage at diagnosis
National Cancer Institute. SEER Cancer Stat Facts: Pancreatic Cancer. Surveillance, Epidemiology, and End Results Program (SEER 21, 2016–2022). Available at: https://seer.cancer.gov/statfacts/html/pancreas.html
American Cancer Society. Cancer Facts & Figures 2026. Atlanta: American Cancer Society; 2026.
Pancreatic Cancer Action Network. Pancreatic Cancer Survival Rate (updated January 2026). Available at: https://pancan.org/facing-pancreatic-cancer/about-pancreatic-cancer/survival-rate/
Khalaf N, El-Serag HB, Abrams HR, Thrift AP. Burden of Pancreatic Cancer: From Epidemiology to Practice. Clin Gastroenterol Hepatol. 2021;19(5):876-884.
Cancer biology, KRAS, and tumor microenvironment
Hosein AN, Brekken RA, Maitra A. Pancreatic cancer stroma: an update on therapeutic targeting strategies. Nat Rev Gastroenterol Hepatol. 2020;17(8):487-505.
Buscail L, Bournet B, Cordelier P. Role of oncogenic KRAS in the diagnosis, prognosis and treatment of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2020;17(3):153-168.
New treatments — daraxonrasib and elraglusib
Revolution Medicines. Daraxonrasib Demonstrates Unprecedented Overall Survival Benefit in Pivotal Phase 3 RASolute 302 Clinical Trial in Patients with Metastatic Pancreatic Cancer. Press release, April 13, 2026. ClinicalTrials.gov: NCT06625320. Available at: https://ir.revmed.com/news-releases/news-release-details/daraxonrasib-demonstrates-unprecedented-overall-survival-benefit
Mahalingam D, Shroff RT, Carneiro BA, et al. Elraglusib and chemotherapy in metastatic pancreatic ductal adenocarcinoma: a randomized controlled phase 2 trial. Nat Med. Published online April 14, 2026. DOI: 10.1038/s41591-026-04327-4. ClinicalTrials.gov: NCT03678883.
Risk factors
American Cancer Society. Pancreatic Cancer Risk Factors. Available at: https://www.cancer.org/cancer/types/pancreatic-cancer/causes-risks-prevention/risk-factors.html
Pancreatic Cancer Action Network. Pancreatic Cancer Risk Factors. Available at: https://pancan.org/facing-pancreatic-cancer/about-pancreatic-cancer/risk-factors/
Park M, Maeda T, Park J-H, et al. Excess pancreatic cancer risk due to smoking and modifying effect of quitting smoking: The Multiethnic Cohort Study. Cancer Epidemiol. 2024;88:102516.
Setiawan VW, Stram DO, Porcel J, et al. Pancreatic cancer following incident diabetes in African Americans and Latinos: The Multiethnic Cohort. J Natl Cancer Inst. 2019;111(1):27-33.
Klein AP. Genetic susceptibility to pancreatic cancer. Mol Carcinog. 2012;51(1):14-24.
Hu C, Hart SN, Polley EC, et al. Association between inherited germline mutations in cancer predisposition genes and risk of pancreatic cancer. JAMA. 2018;319(23):2401-2409.
Racial disparities
Tavakkoli A, Singal AG, Waljee AK, et al. Racial Disparities and Trends in Pancreatic Cancer Incidence and Mortality in the United States. Clin Gastroenterol Hepatol. 2020;18(1):171-178.
Khawja SN, Mohammed S, Silberfein EJ, et al. Pancreatic cancer disparities in African Americans. Pancreas. 2015;44(4):522-527.
New-onset diabetes as early signal
Sharma A, Kandlakunta H, Nagpal SJS, et al. Model to determine risk of pancreatic cancer in patients with new-onset diabetes. Gastroenterology. 2018;155(3):730-739. (ENDPAC score)
Pannala R, Leirness JB, Bamlet WR, et al. Prevalence and clinical profile of pancreatic cancer-associated diabetes mellitus. Gastroenterology. 2008;134(4):981-987.
Surveillance in high-risk individuals (CAPS)
Blackford AL, Canto MI, Dbouk M, et al. Pancreatic Cancer Surveillance and Survival of High-Risk Individuals. JAMA Oncol. 2024;10(8):1087-1096. DOI: 10.1001/jamaoncol.2024.1930.
Dbouk M, Katona BW, Brand RE, et al. The Multicenter Cancer of Pancreas Screening Study: Impact on Stage and Survival. J Clin Oncol. 2022;40(28):3257-3266.
Goggins M, Overbeek KA, Brand R, et al. Management of patients with increased risk for familial pancreatic cancer: updated recommendations from the International Cancer of the Pancreas Screening (CAPS) Consortium. Gut. 2020;69(1):7-17.
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Genetic/Familial High-Risk Assessment: Breast, Ovarian, Pancreatic, and Prostate. (Most recent version, accessed via NCCN.org)
Screening limitations and CA 19-9
Owens DK, Davidson KW, Krist AH, et al; US Preventive Services Task Force. Screening for Pancreatic Cancer: US Preventive Services Task Force Reaffirmation Recommendation Statement. JAMA. 2019;322(5):438-444.
Ballehaninna UK, Chamberlain RS. The clinical utility of serum CA 19-9 in the diagnosis, prognosis and management of pancreatic adenocarcinoma: An evidence based appraisal. J Gastrointest Oncol. 2012;3(2):105-119.
Joy is the founder and executive director of Proactive Health Labs (pH Labs), a national nonprofit focused on holistic health education. A certified holistic wellness coach with 15 years of practice and 40 years as a healthcare attorney, she writes about the science of staying well from a perspective that is both rigorous and personal — including her own experience navigating genetic risk, surveillance, and the questions every patient deserves to have answered.